ScanAI Platform
ScanAI Digital Pathology Platform
AI-powered digital pathology for accurate cervical cancer diagnosis
ScanAI Advantages over Conventional Methods
ScanAI by Scanome is a groundbreaking digital pathology platform designed to accurately diagnose cervical cancer. Leveraging state-of-the-art artificial intelligence, machine learning, and advanced image analysis, ScanAI transforms the way cancer diagnosis is approached and accelerates the path to life-saving treatments.
You can accelerate your diagnosis today at https://scanai.scanome.com/. Reach out to contact@scanome.com for login credentials. Currently, our platform is open for research use.
Enhanced Accuracy
Our advanced AI algorithm has been meticulously designed to analyze and interpret cervical cell samples with an accuracy rate that surpasses conventional methods.
Faster Results
ScanAI dramatically reduces the time required for analyzing samples, providing healthcare professionals with quicker results.
Enhanced Patient Care
ScanAI empowers healthcare professionals to provide better patient care by delivering accurate and timely results.
Cost Effective
By automating the cervical cancer screening process, ScanAI not only saves time but also reduces the overall cost of screening.
Global Accessibility
ScanAI is designed to work seamlessly with various screening methods and medical equipment, making it easily accessible to healthcare providers worldwide.
Key Features of ScanAI
Comprehensive AI-powered capabilities for modern digital pathology
ScanAI is specifically developed to identify and diagnose cervical cancer, automating one of the most popular digital pathology tests.
Our sophisticated AI algorithms and machine learning models analyze cell samples with remarkable precision and speed, identifying subtle patterns and abnormalities.
ScanAI provides detailed, easy-to-understand reports, including diagnostic results, biomarker quantification, and risk assessment.
ScanAI is designed to integrate effortlessly with existing laboratory information systems (LIS) and digital pathology workflows.
We are committed to staying at the forefront of AI advancements. ScanAI is continuously updated to meet the evolving needs of the medical community.
ScanAI adheres to the highest industry standards for data security and privacy, ensuring your sensitive patient data is protected.
Transforming the Cancer Diagnosis Workflow
By harnessing the power of artificial intelligence and machine learning, ScanAI empowers pathologists and clinicians to deliver rapid, accurate, and reliable diagnoses for cervical cancer. Our advanced digital pathology software is designed to enhance diagnostic capabilities, streamline workflows, and ultimately improve patient outcomes.
Prioritize Cases
Quickly identify high-risk or complex cases that require immediate attention, enabling pathologists to allocate their time and resources more effectively.
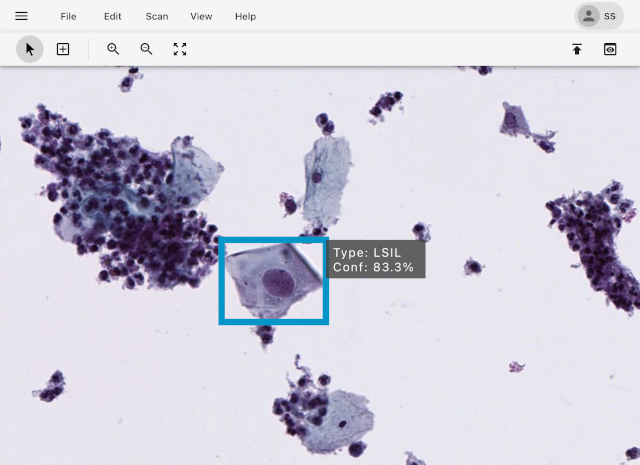
Highlight Anomalous Cells
Advanced AI algorithms accurately detect and highlight anomalous cells according to the Bethesda Reporting System.
Second Opinion for Extra Confidence
Provides an automated read on every slide, offering pathologists an additional layer of confidence in their diagnoses.
Decrease Diagnosis Time
Significantly reduces the time necessary for diagnosis by automating the initial analysis of Pap smear images.
Audit Trail for Diagnosis Decision
Maintains a comprehensive audit trail for each diagnosis decision, allowing for increased transparency and traceability.
Collaboration Among Pathologists
Digital platform allows multiple pathologists to work on the same slide remotely and simultaneously, facilitating collaboration.
Ready to Transform Your Diagnostic Workflow?
Reach out to contact@scanome.com if you would like to test our ScanAI platform in your laboratory! Currently, our platform is open for research use. We are working towards having our platform certified for medical use with CE-IVD, ISO 13485 and ISO 9001.
